b-AP15 NLT 98%
Product Number : MC501438
CAS Number : 1009817-63-3
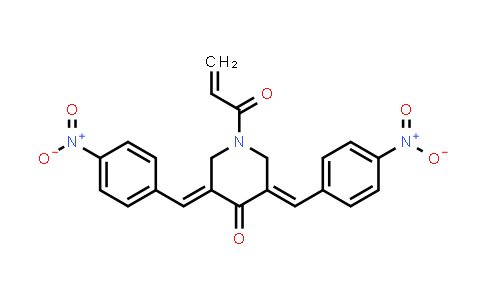
Molecular Formula : C22H17N3O6 | Molecular Weight : 419.39
Synonyms : NSC 687852;(3E,5E)-1-Acryloyl-3,5-bis(4-nitrobenzylidene)piperidin-4-one
Quote Request| Purity | NLT 98% |
|---|---|
| Storage | at 20ºC 2 years |
| MolCore specializes in manufacturing high-purity CAS No.1009817-63-3, b-AP15 with the molecular formula C22H17N3O6 and molecular weight 419.39 delivering critical API intermediates for global pharmaceutical and research industries, certified under ISO quality systems. | |
* The above information is for reference only.
| Chemical Name | b-AP15 |
|---|---|
| CAS Number | 1009817-63-3 |
| Molecular Formula | C22H17N3O6 |
| Molecular Weight | 419.39 |
| Synonyms | NSC 687852;(3E,5E)-1-Acryloyl-3,5-bis(4-nitrobenzylidene)piperidin-4-one |
b-AP15 is a specific inhibitor of the deubiquitinating enzymes UCHL5 and Usp14. IC50 & Target: UCHL5/Usp14[1] In Vitro: Purified 19S proteasomes (5 nM) are treated with indicated concentrations of b-AP15 and DUB activity is determined by detectionof Ub-AMC cleavage. The IC50 value (2.1±0.411 μM) is determined from log concentration curves in Graph Pad Prism using non linear regression analysis. b-AP15 as a previously unidentified class of proteasome inhibitor that abrogates the deubiquitinating activity of the 19S regulatory particle. b-AP15 inhibited the activity of two 19S regulatory-particle-associated deubiquitinases, ubiquitin C-terminal hydrolase 5 (UCHL5) and ubiquitin-specific peptidase 14 (USP14), resulting in accumulation of polyubiquitin. b-AP15 induced tumor cell apoptosis that is insensitive to TP53 status and overexpression of the apoptosis inhibitor BCL2[1]. The ability of b-AP15 is determined to inhibit proteasome deubiquitinase activity using Ub-AMC as the substrate. An IC50 of 16.8±2.8 μM is observed[2]. b-AP15 is a specific USP14 and UCHL5 inhibitor, which blocks growth and induces apoptosis in MM cells[3]. In Vivo: b-AP15 (2.5 mg/kg) inhibits tumor growth in syngenic mice models with less frequent administration schedules. We administered b-AP15 to C57BL/6J mice with Lewis lung carcinomas (LLCs) using a 2-d-on, 2-d-off schedule and to BALB/c mice with orthotopic breast carcinoma (4T1) using a 1-d-on, 3-d-off schedule. b-AP15 significantly inhibited tumor growth in both models, with T/C=0.16 (P≤0.01) for the C57BL/6J mice and T/C=0.25 (P≤0.001) for the BALB/c mice. A reduction in the number of pulmonary metastases also is observed in the group of mice with 4T1 breast carcinomas treated with b-AP15[1].
© Copyright 2015-2025 Hangzhou MolCore BioPharmatech Co.,Ltd. All rights reserved.
