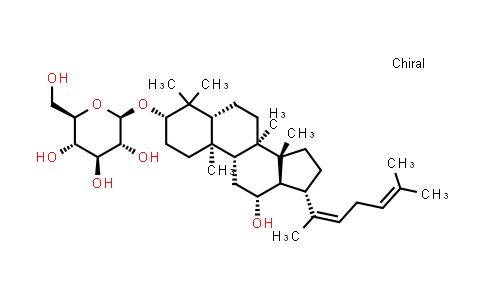
Ginsenoside Rh3 NLT 98%
Product Number : MC504457
CAS Number : 105558-26-7
Molecular Formula : C36H60O7 | Molecular Weight : 604.86
Quote Request| Purity | NLT 98% |
|---|---|
| Storage | at 20ºC 2 years |
| MolCore specializes in manufacturing high-purity CAS No.105558-26-7, Ginsenoside Rh3 with the molecular formula C36H60O7 and molecular weight 604.86 delivering critical API intermediates for global pharmaceutical and research industries, certified under ISO quality systems. | |
* The above information is for reference only.
| Chemical Name | Ginsenoside Rh3 |
|---|---|
| CAS Number | 105558-26-7 |
| MDL Number | MFCD10566602 |
| Molecular Formula | C36H60O7 |
| Molecular Weight | 604.86 |
Ginsenoside Rh3 is a bacterial metabolite of Ginsenoside Rg5. Ginsenoside Rh3 treatment in human retinal cells induces Nrf2 activation. IC50 & Target: Nrf2[1] In Vitro: Ginsenoside Rh3 inhibits UV-induced oxidative damages in retinal cells via activating nuclear-factor-E2-related factor 2 (Nrf2) signaling. Ginsenoside Rh3 treatment in retinal cells induces Nrf2 activation. The potential activity of Ginsenoside Rh3 is tested on Nrf2 signaling in the retinal pigment epithelium cells (RPEs). The qRT-PCR assay results demonstrate that treatment with Ginsenoside Rh3 dose-dependently increases mRNA transcription and expression of key Nrf2-regulated genes, including HO1, NQO1 and GCLC. Consequently, protein expressions of these Nrf2-dependent genes (HO1, NQO1 and GCLC) are also significantly increased in Ginsenoside Rh3 (3-10 μM)-treated RPEs. Notably, although Nrf2 mRNA level is unchanged after Ginsenoside Rh3 treatment, its protein level is significantly increased by Rh3[1]. EZ-Cytox assay is used to assess the effect of ginsenoside-Rh3 on SP 1-keratinocytes viability. Ginsenoside Rh3 (0.01, 0.1, 1 and 10 μM) shows no cytotoxic effect at all concentrations[2]. In Vivo: The potential effect of Ginsenoside Rh3 is examined on mouse retina, using the light-induced retinal damage model. Ginsenoside Rh3 intravitreal injection (5 mg/kg body weight, 30 min pre-treatment) significantly attenuates light-induced decrease of both a- and b-wave amplitude. The electroretinography (ERG)'s a-wave decreases to 46.03±1.62% % of control level after light exposure, which is back to 71.84±7.51% with Ginsenoside Rh3 administration. The b-wave is 40.19±3.34% of control level by light exposure, and Rh3 intravitreal injection brings back to 80.01±2.37% of control level[1].
© Copyright 2015-2025 Hangzhou MolCore BioPharmatech Co.,Ltd. All rights reserved.
